 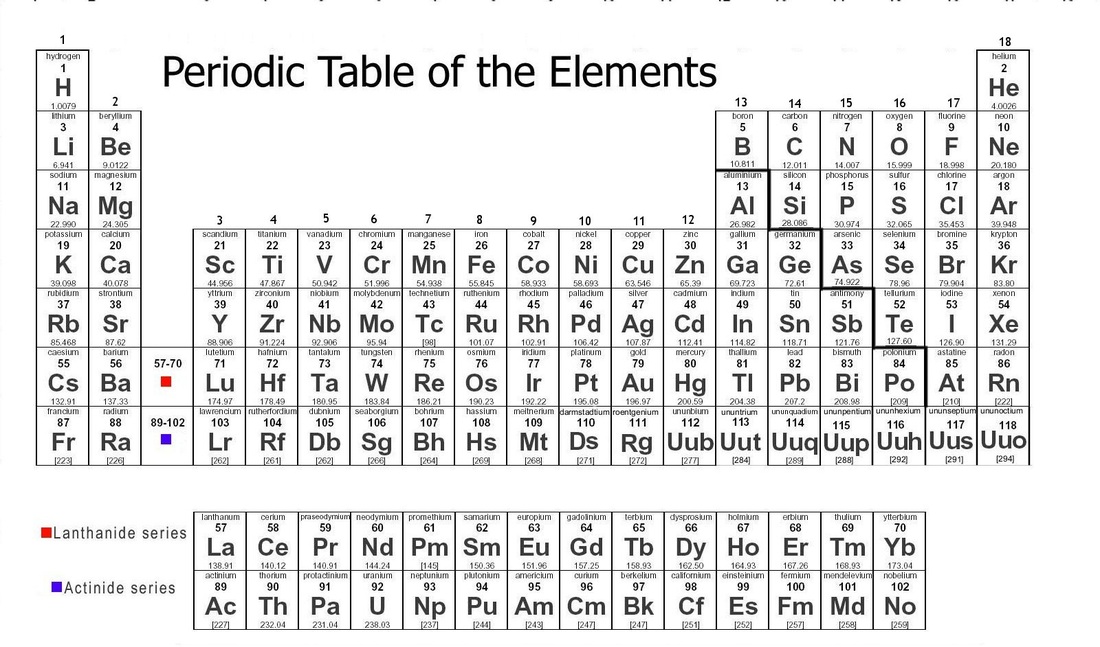
Also, the California Department of Education recently published a set of equations and constants as a resource for our new CAST (California Science Test). I recently decided that the old CA State table we used for years needed to go away. When you are all done creating your masterpiece, you can download it as a pdf file. You can also customize font size and add a logo. Atomic mass, electron configuration, density, boiling point, freezing point - all can be included by simply checking a box. This is online software that allows an amazing amount of customization, from color schemes to the data that you want displayed. This is the second "Build Your Own Table" website to which I have linked. If the file does not reside on my server, you will be directed to a page where the table CAN be downloaded. Only seven of these tables are my creation all rights are retained by the original authors of the others.Ĭlick on the element symbol to download the PDF file. The quality of the results will depend in large part on the quality of your printer. All of the tables will print on a single 8½ x 11 page, though some may require color to retain their appearance. The following descriptions include a sample element from each periodic table. The program can be downloaded from the Adobe Website. To view and print these files, you will need to install the free Adobe Acrobat Reader program on your computer. Unfortunately, the relative nature of these suffixes mandates that the ion formula/ion name combinations of the polyatomic ions must simply be memorized.Unless otherwise indicated, the following tables are in Acrobat PDF format. 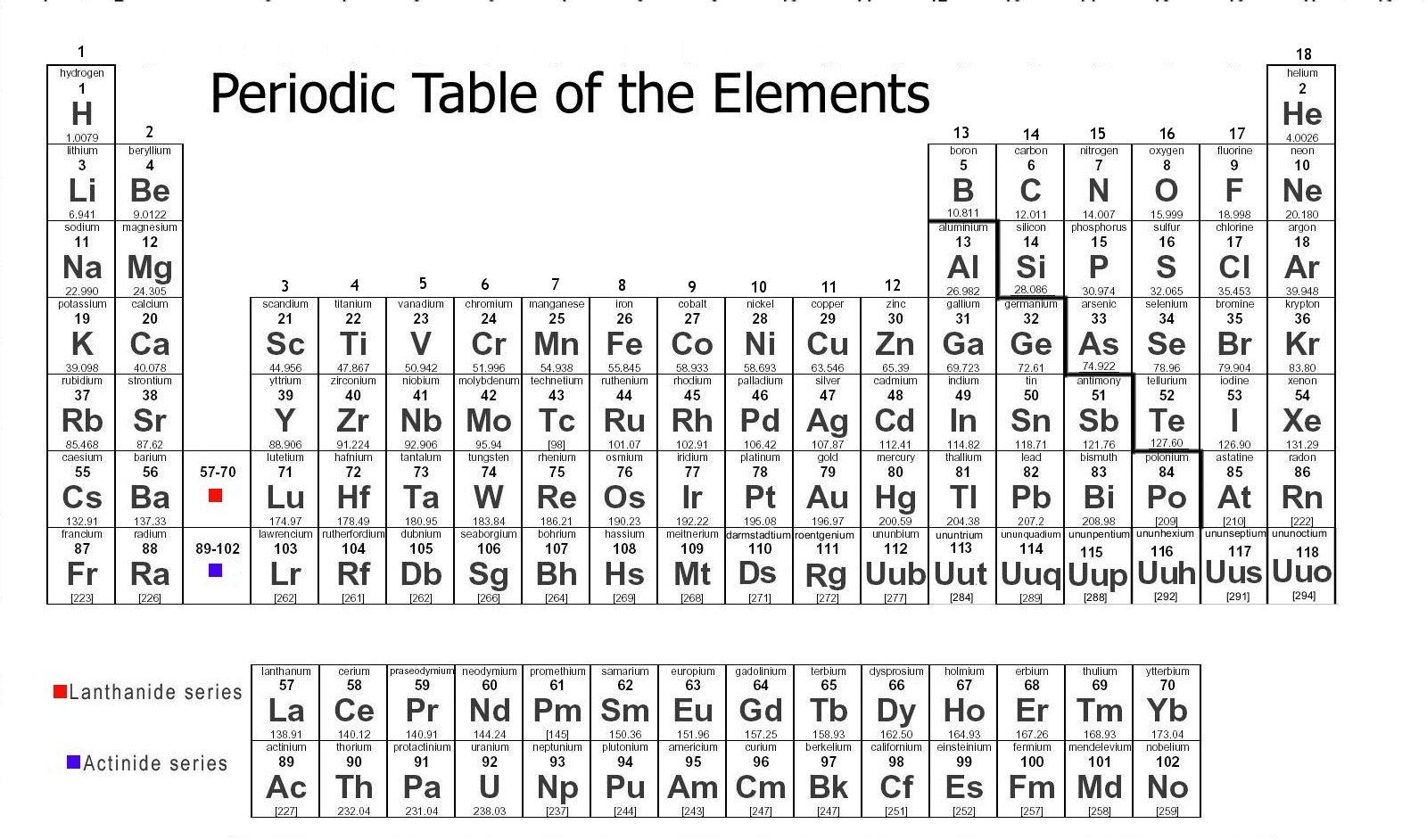 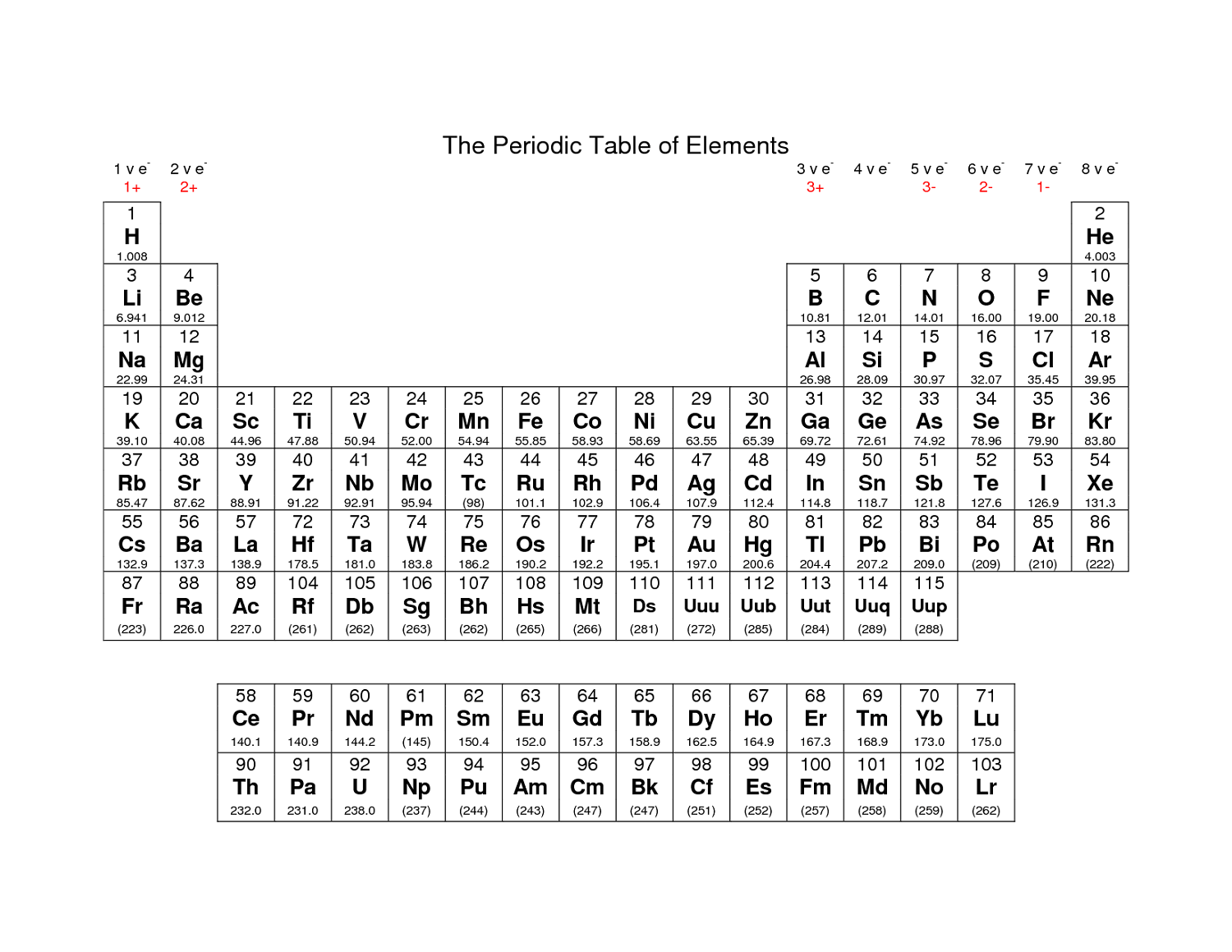
Additionally, both the nitr ate ion and the sulf ite ion contain three oxygens, but these polyatomic ions do not share a common suffix. For example, both the nitrate ion, symbolized as NO 3 −, and the sulfate ion, symbolized as SO 4 2 −, share an " -ate" suffix, however, the former contains three oxygens, and the latter contains four. Note that all of the polyatomic ions whose names end in " -ate" contain one more oxygen than those polyatomic anions whose names end in " -ite." Unfortunately, much like the common system for naming transition metals, these suffixes only indicate the relative number of oxygens that are contained within the polyatomic ions. Additionally, these suffixes also indicate the relative number of oxygens that are contained within the polyatomic ions. Therefore, " - ate" and " - ite" suffixes are employed, in order to denote that the corresponding polyatomic ions are part of a series. A single suffix, "-ide," is insufficient for distinguishing the names of the anions in a related polyatomic series. The remaining polyatomic anions, which all contain oxygen, in combination with another non-metal, exist as part of a series in which the number of oxygens within the polyatomic unit can vary. However, only two of these, the hydroxide ion and the cyanide ion, are named using the "- ide" suffix that is typically indicative of negatively-charged ions. Note that only two polyatomic ions in this table are cations, hydronium ion (H 3O +) and ammonium ion (NH 4 +), the remaining polyatomic ions are all negatively-charged and, therefore, are classified as anions. \): Common Polyatomic Ion Names and Formulas Ion Name 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
